Designing a Robotic Vaccine Storage and Delivery System for Controlled Environments

Client Need & Project Overview
Maintaining proper vaccine storage conditions has always been a critical challenge, but historically, most systems relied on manual handling, limited monitoring, and fragmented workflows between refrigeration and access.
TruMed set out to rethink this model by combining controlled storage with automated handling. The goal was to create a system that could not only maintain strict temperature requirements, but also improve how vaccines were stored, accessed, and delivered within that environment.
NOVO was selected to design a custom refrigeration and robotic delivery system that integrated temperature-controlled storage with automated movement between compartments. To our knowledge, this was the first product to use a robot to traverse between a refrigerator and freezer within a single system.
The system combined refrigeration, robotics, sensing, power management, and cloud connectivity into a single integrated platform, aligning closely with today’s connected and automated healthcare systems.
Key Engineering Challenges & Solutions
| Challenge | Solution |
|---|---|
| Maintaining 2–8°C across the entire system volume (not just localized zones) | Designed a custom refrigeration system that maintains uniform temperature across all storage locations, even during active operation |
| Robotic movement between freezer and refrigerator compartments | Developed a robot that traverses both environments while preventing air mixing and preserving thermal stability |
| Temperature control during power outages (12-hour holdover requirement) | Engineered the system to maintain full temperature specifications on battery power, requiring careful power management and defrost system innovation |
| Storage and retrieval across varied vaccine formats | Developed robotic handling and control systems capable of reliably managing a wide range of packaging types |
Technical Innovations & System Design
Refrigeration and Thermal Control
The core of the system was a custom refrigeration architecture designed to maintain a tightly controlled 2–8°C range across the entire internal volume. This requirement had to be met during active operation, across varying ambient conditions, and even during power outage scenarios.
Significant effort was also required in sealing and insulation design to improve power efficiency and control condensation and ice formation.
Robotic System Design
NOVO designed a robotic delivery system capable of traversing between refrigerated and frozen compartments. A key innovation was enabling this movement without introducing large volumes of freezer air into the refrigerator, preserving system stability.
Ensuring reliable performance across a wide range of vaccine packaging formats required careful integration of mechanical design and control systems.
Sensing and System Reliability
Sensor networks for temperature, position, and presence were required to operate reliably in cold environments subject to icing, adding complexity to both hardware selection and system design.
Connectivity and Workflow Integration
The system incorporated cloud connectivity with Ethernet as the primary connection and WiFi and LTE as backup options. This enabled monitoring, alerting, and workflow integration, including inventory tracking, barcode scanning, and EMR connectivity.
Validation & Testing
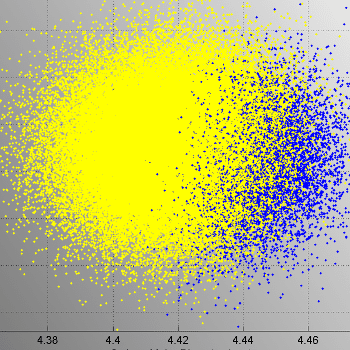
NOVO conducted extensive computational fluid dynamics (CFD) modeling to evaluate temperature uniformity across all storage locations. A new test method for refrigeration systems was developed and later sanctioned by the CDC and ASTM.
This approach enabled precise measurement of temperature distribution, airflow, and air leakage, ensuring the system performed as intended.
Results & Impact
The final system delivered a first-of-its-kind solution that combined precise temperature control, robotic automation, and connected system capabilities. It improved workflow efficiency, reduced vaccine waste, and enabled small medical practices to confidently manage vaccine inventory.
Why NOVO Engineering?
This project demonstrates NOVO’s ability to solve complex, multidisciplinary challenges across thermal engineering, robotics, controls, and system integration.
By bringing these domains together into a single platform, NOVO anticipated trends that now define modern healthcare systems, including automation, connected monitoring, and integrated cold chain solutions.
Let’s Talk About Your Project
If you are developing a system that combines environmental control, automation, and connectivity, NOVO Engineering can help.
Let’s start a conversation about your goals.