Engineering a Seamless Diabetes Experience
Client Need & Project Overview
Today, connected medical devices are expected to simplify treatment decisions and support real-time data sharing. When Bigfoot Biomedical began developing the Unity™ Diabetes Management System in 2021, those expectations were just beginning to take shape.
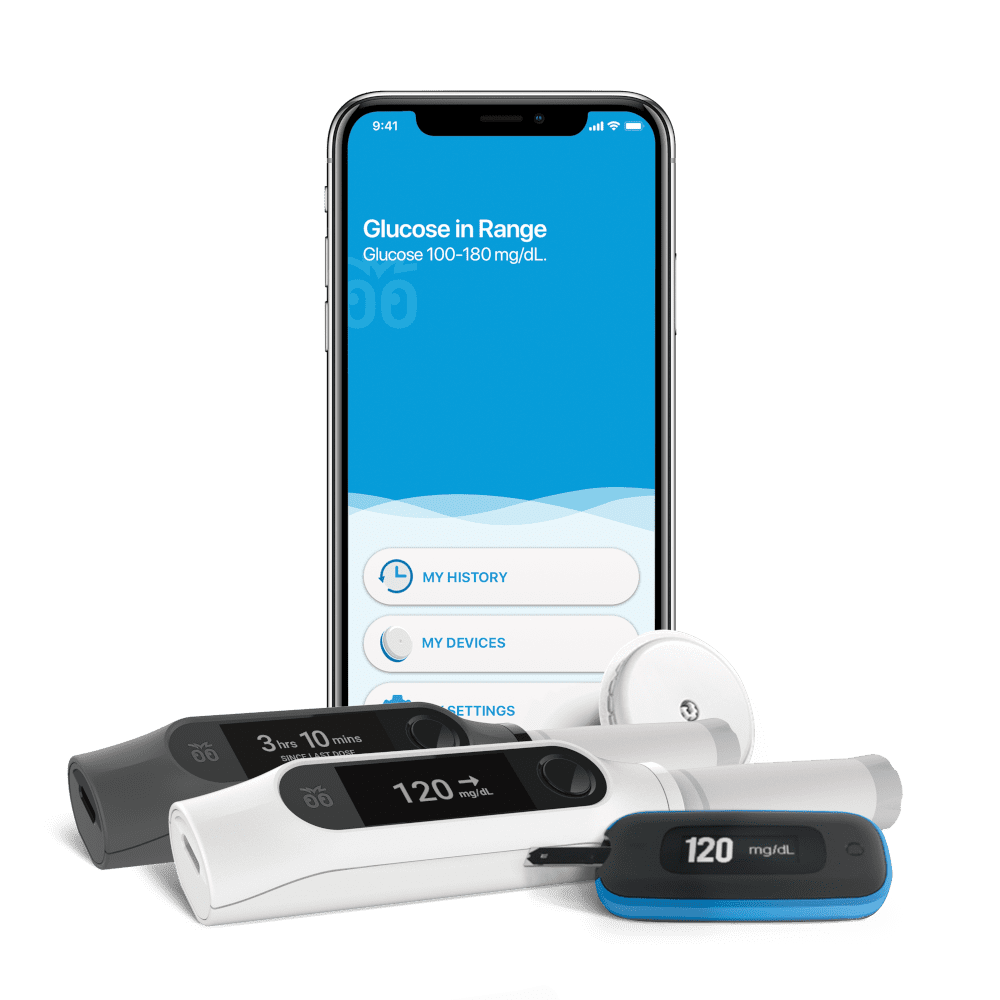
Unity was built around familiar tools such as disposable insulin pens, continuous glucose monitors, and smartphones to provide an intuitive, supportive insulin dosing experience. As Bigfoot puts it, the system helps answer a critical daily question: “How much insulin would my doctor recommend I take right now?”
To bring this vision to life, Bigfoot partnered with NOVO Engineering. Our team developed the embedded firmware, wireless communication, hardware integration, and test infrastructure needed to deliver a secure, FDA-cleared product.
The Unity system features smart pen caps compatible with common rapid- and long-acting insulin pens. It connects with Abbott’s FreeStyle Libre 2 integrated continuous glucose monitor (iCGM), a blood glucose meter (BGM), and a mobile app to provide dose guidance, data sharing, and a more manageable experience for people with diabetes.
Key Engineering Challenges & Solutions
| Challenge | NOVO’s Solution |
|---|---|
| Secure, multi-deviceconnectivity | Developed firmware and system architecture for wireless communication across iCGM, BGM, and mobile platforms, balancing low power use, compliance, and ease of use |
| System integration | Delivered electromechanical and software designs compatible with multiple insulin pens, enabling accurate dose tracking and seamless user experience |
| End-to-end security | Built a security framework from device to cloud with encrypted wireless protocols and authenticated communication |
| Test infrastructure at scale | Designed custom automated test equipment (ATE) to monitor performance and support rapid iteration and verification |

Technical Innovations & System Design
Embedded Systems & Wireless Protocols
NOVO developed embedded firmware and communication protocols allowing Unity pen caps to connect with the FreeStyle Libre 2 iCGM, a BGM, and mobile app. The system supported encrypted Bluetooth and other wireless standards for secure, real-time data exchange.
Cross-Compatible Hardware Design
Bigfoot needed a hardware solution for various FDA-approved insulin pens. NOVO engineered a form factor and sensing system that fit securely, tracked doses accurately, and met ergonomic needs.
Test Automation to Accelerate Development
NOVO deployed ATE platforms to monitor functional parameters across configurations. This early investment enabled faster iteration, higher reliability, and simplified verification.
Intellectual Property Contributions
NOVO’s engineering work contributed to several patents covering connectivity, user interaction, and embedded intelligence:
These patents reflect NOVO’s role in developing foundational systems that now define connected insulin delivery.

Results & Impact
- Regulatory success: FDA 510(k) clearance for a novel smart pen cap system
- Simplified management: Personalized insulin guidance supported by iCGM, BGM, and mobile application
- Seamless data sharing: Secure transmission of glucose data to physicians
- End-to-end security by design: Chain of trust from device to cloud to protect patient data and product integrity
- Documented innovation: Patents recognizing NOVO’s engineering contributions to smarter insulin delivery
Why NOVO Engineering?
Bigfoot Biomedical brought the vision. NOVO delivered the engineering to make it real.
From embedded systems to secure communication and scalable testing, NOVO helped transform a complex product idea into a connected device aligned with where the medtech market is now headed.
Let’s build your next connected medical device
Reach out to NOVO Engineering to explore how we can help bring your innovation to life.







