The manifesto for Agile Development reads:
Individuals and interactions over processes and tools
Working software over comprehensive documentation
Customer collaboration over contract negotiation
Responding to change over following a plan
How does this philosophy, which was originally developed for software, apply to medical device development – especially in regards to following regulations such as ISO 13485 and Title 21 CFR Part 820?
The goals and values of Agile and medical device development are actually generally well-aligned. The specific practices of Agile development can be tailored to meet the demands of medical device development with a focus on safety, reliability, and security.
MEDICAL DEVICE REGULATORY GOALS AND VALUES
The traditional approach to medical device development is the Waterfall development process – a sequential process that flows from one step to another once a step is fully complete. However, neither ISO 13485 nor 21 CFR 820 require a specific process for development. Instead, the focus of these regulations is on the requirements for the manufacturer’s quality management system (QMS) and the attendant design controls used during development. Regulators are primarily concerned with ensuring that a development process is established and followed and that process ensures the safety, reliability, efficacy, and security of the resulting medical device.
Regulators have shown a growing acceptance of Agile methodologies in the medical device development process. Although no guidance is yet available from the FDA, industry groups like the Association for the Advancement of Medical Instrumentation (AAMI) have published guidance documents [1] showing that the goals and values of Agile and medical device development are more aligned than many people believe.
AGILE GOALS AND VALUES
As originally conceived in the Agile Manifesto [2], Agile doesn’t constitute a set of practices and processes. Instead, Agile is a statement of values and goals specifically for the software development process. The goal of Agile is to optimize the process of development for quality, productivity, predictability, and effectiveness. The four core values of Agile are:
- Individuals and interactions over processes and tools.
- Working software over comprehensive documentation.
- Customer collaboration over contract negotiation.
- Responding to change over following a plan.
The core philosophy of Agile strives to encourage a high degree of input and collaboration between the client and the company with whom they’ve chosen to work. It focuses on breaking down the development process into iterative sprints to accurately estimate costs and set clear predictable timelines. Other focuses of Agile are flexibility, regular testing in the development process to ensure quality, reduced risk, and faster ROI. Ultimately, the goal of Agile is to facilitate a stronger, more effective, and efficient working relationship between the client and the product development team.
In practice, because the manifesto doesn’t attempt to provide a definition of specific development practices, one or more different Agile frameworks are used – examples include Scrum, Extreme Programming, Kanban, and feature-driven development. While the appeal of Agile spans engineering disciplines, no specific frameworks have been published that apply Agile principles to anything other than software (per the above). Despite this, all development projects can benefit from a set of practices drawn from various sources that are intended specifically to meet the goals and values of Agile.
What Agile Isn’t: A Complete Absence of Documentation
Agile is often misunderstood to promote development practices that are undisciplined. In particular, there appears to be an impression that Agile advocates for the complete absence of documentation. This misunderstanding is likely due in part to the way in which the Agile Manifesto expresses its values. And unfortunately, it’s reinforced by the poor implementation of Agile in certain industries. The value statement “Working software over comprehensive documentation” isn’t intended to mean that documentation has no value. It’s actually an expression that the quality of the actual product is more important than the volume of the documentation for the product. This statement has been misused to suggest that documentation isn’t important in an attempt to avoid a task seen as less desirable or exciting.
The True Goal of Agile
The true goal of Agile is to promote development efficiency while delivering a product that meets the needs of the customer. In the case of medical device development, the needs of the customer include providing a product that has a documented ability to meet the regulatory goals of safety, reliability, efficacy, and security.
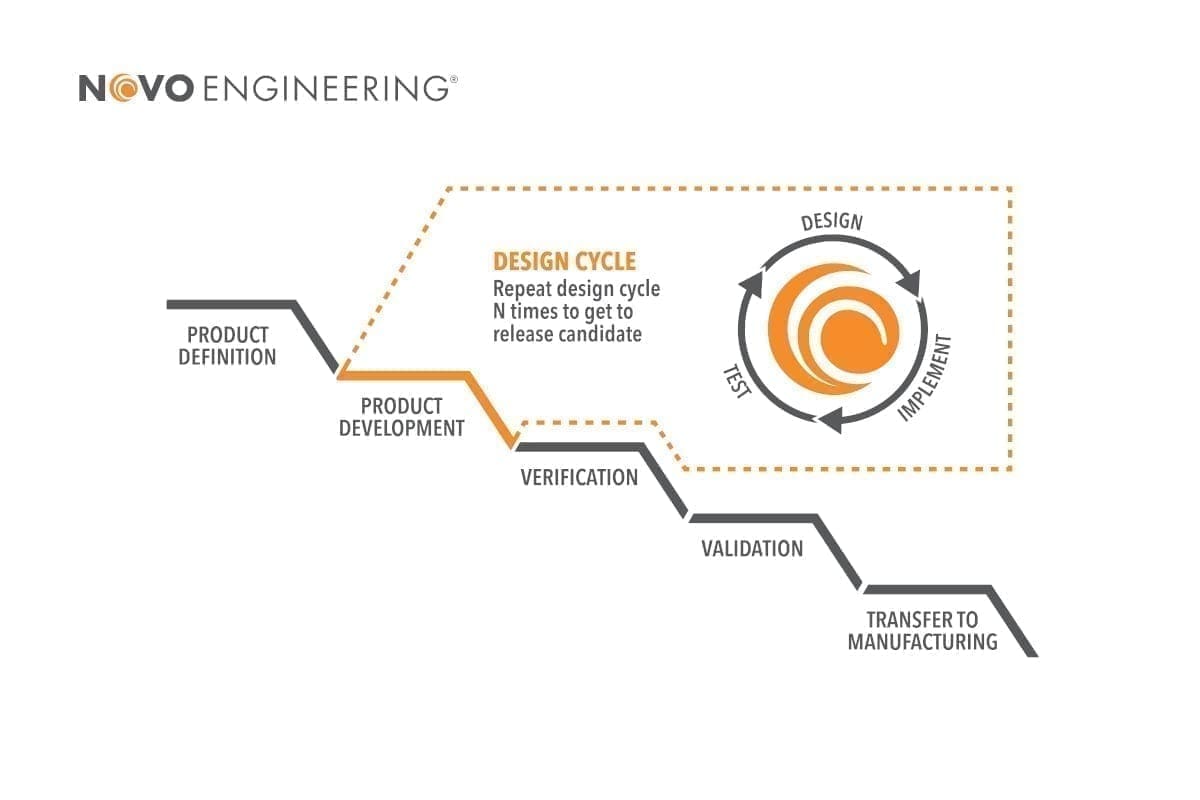
NOVO’S MEDICAL DEVICE DEVELOPMENT PROCESS
NOVO’s quality management system is designed for our business model and our role in the development phase of the product lifecycle. Our product development process has been refined through many successful projects to meet the needs of the types of multi-disciplinary development projects that we specialize in, including medical device development. The overall process includes a phased approach to medical device development that is consistent with a traditional Waterfall plan. However, we tailor the development plan for each phase of development, using the best practices that are suited to each engineering discipline and to the specific needs of the project. This can include using the Agile methodology. In all cases, NOVO seeks to maximize the value we provide to our clients by optimizing our medical device development process for the clients’ specific goals.
We serve many different clients in different industries with different needs. It’s critical to our business to be able to tailor the product development plan for each project. The one need that’s common to all clients is the need to deliver a high-quality product as efficiently as possible that also satisfies the requirements and needs of all stakeholders. In this sense, the goals of Agile are very well aligned with the needs of our clients, whether the product is a medical device or another type of product.
If you’re seeking a partner that will assist in the entire medical device development process lifecycle, from inception to pre-production manufacturing, NOVO is here to assist you – using whatever development methods best fit your goals. With 60+ engineers with an average of 16 years of experience recruited from multiple industries, we have the expertise to deliver an innovative and efficient process for product development.
[1] AAMI TIR45 Guidance on the use of AGILE practices in the development of medical device software
[2] Agile Manifesto. Retrieved May 24, 2019, from https://agilemanifesto.org/.
